 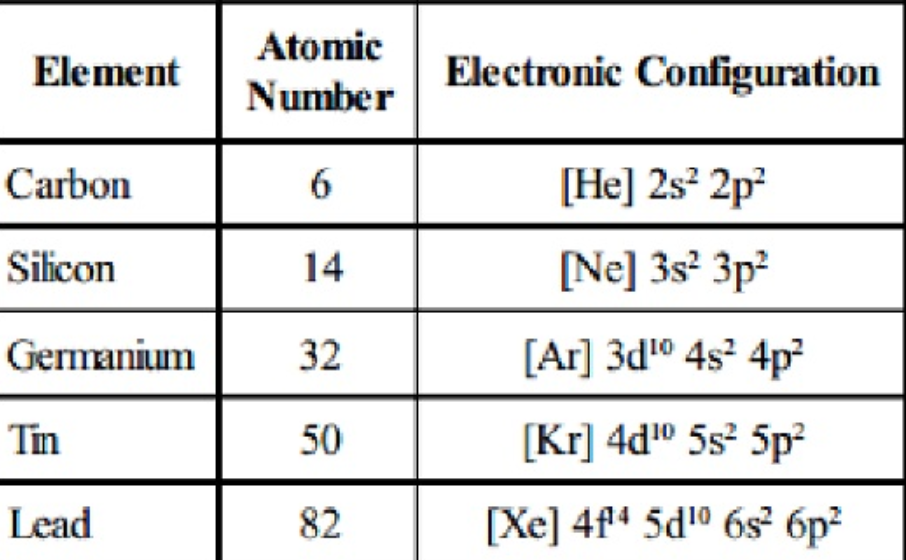
Mass number – the number of protons and neutrons in the nucleus of an atom.Īs the outer shell electrons get further away from the nucleus, this force of attraction becomes weaker. Key termsĪtomic number – the number of protons in an atom. This strong force of attractions between the electrons and the nucleus holds them in place. As electrons are negatively charged, they are attracted strongly to the positive charge of the nucleus. The nucleus is positively charged due to the presence of the protons. For example, group 7 elements have 7 electrons in their outer shell. All elements in the same group have the same number of electrons in their outer energy shell. The group number of an element tells you how many electrons there are in the outer shell and the period number tells you how many electron-containing energy shells the element has. The periodic table in terms of electronic structure To determine the number of neutrons we use the equation: Carbon has a mass number of 12 (so there are 12 protons and neutrons) and an atomic number of 6 (so there are 6 protons). This also means that carbon must have 6 electrons. 
Using carbon as an example, we known that it has an atomic number of 6 so has 6 protons. Therefore, if we know the mass number and atomic number of an element, we can work out the numbers of protons, neutrons and electrons it has. The number of protons and electrons in an atom is always equal, as a proton has a charge of +1 and an electron has a charge of -1 and the atom must have an overall neutral charge. The number of neutrons present in the nucleus of the atom can be deduced by subtracting the number of protons from the total number of protons and neutrons as shown in the equation: The chemical elements in the periodic table are arranged in order of increasing atomic number. Atomic number (proton number) tells you the number of protons present in the nucleus of the atom. The lower of the two numbers is the atomic number. Mass number tells you the total number of protons and neutrons in the nucleus of the atom. The higher of the two numbers is the mass number. This also allows you to predict properties of other elements in that group. Their chemical reactivity therefore depends on the number of electrons they need to gain or lose to achieve this full outer shell.Īs elements in the same group will want to gain or lose the same number of electrons to gain a full outer shell, this explains why elements in the same group have similar chemical properties. This means that all elements in the same group have the same number of electrons in their outer energy shell.Ītoms always react to gain or lose electrons in order to have a full outer shell. The group number of the element gives you the number of electrons in the outer shell. Why elements in the same group of the periodic table have similar properties The elements in groups 7 and 0 are all non-metals. In group 5 only antimony and bismuth are metals and in group 6 polonium is the only metal. In group 4 carbon and silicon are the only non-metals, with all elements below silicon being metals. In group 3 the only non-metal is boron, with all elements below boron being metals. The metals are found in the middle and on the left side of the periodic table and the non-metals on the right side of the periodic table, as shown in the diagram on the next page.Īll of the elements in groups 1 and 2 are metals. With the exception of carbon and silicon, all non-metals will not conduct electricity, whereas all metals do conduct electricity. Tend to form negative ions and covalent bondsĮxcellent heat and electricity conductorsĭo not tend to conduct electricity (carbon and silicon are exceptions)Ĭontain oxides that react with acids to produce a salt and waterĮlectrical conductivity can be used to classify an element as a metal or non-metal. Tend to have low melting and boiling points Tend to be solids that have high densities and high melting and boiling points The chemical and physical properties of metallic and non-metallic elements differ, as shown in the table below. Approximately 75% of the elements on the periodic table are metals and 25% non-metals. The periodic table is made up of metallic and non-metallic elements. Metals and non-metals in the periodic table Each row in the periodic table is called a period. The vertical columns are known as groups. In the periodic table, elements with similar properties form columns. The periodic table shows all the different elements that are in existence. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
